Home Industry Approvals
INDUSTRY APPROVALS
The usage of Stevia extracts as a sweetener for human consumption has been approved by major regulatory authorities across the world. In 2008 and 2009, the Food and Agriculture Organization, World Health Organization’s Joint Expert Committee on Food Additives (JECFA), a global panel of food ingredient safety experts, and the United States Food and Drug Administration (FDA) stated the use of high purity Steviol glycosides (≥95%) is safe for human consumption, with an acceptable daily Intake (ADI) expressed in Steviol equivalents of up to 4 milligrams per kilogram of body weight per day. In general, stevia is non-toxic, non-mutagenic, non-carcinogenic.
In 2011, the European Commission authorized the use of high purity Steviol glycosides (≥95%) in foods and beverages across the European Union. The Food Safety and Standards Authority of India approved the same in March 2015, while Japan has been using Stevia for over 50 years. Over 150 countries have approved Stevia for human consumption till now.
In India the Stevia Consumption was approved in November, 2015 by FSSAI, FSS act 2006, through a vide gazette notification on 13th November, 2015 F.No P. 15025/208/2013-PA/FSSAI
Below are links to key multiple global regulatory agencies that have approved the use of high purity stevia extracts:
- AgenceFrançaise de Sécurité Sanitaire des Aliments (AFSSA, or French Agency for Food Safety)
- Codex Alimentarius Commission (CAC)
- European Food Safety Authority (EFSA)
- European Commission
- Food Standards Australia New Zealand (FSANZ)
- Health Canada
- Joint FAO/WHO Expert Committee on Food Additives (JECFA)
- U.S. Food and Drug Administration (FDA)
Infographics showing list of countries where Stevia is approved.
WHERE IN THE WORLD IS STEVIA
A MAP OF STEVIA REGULATORY APPROVALS
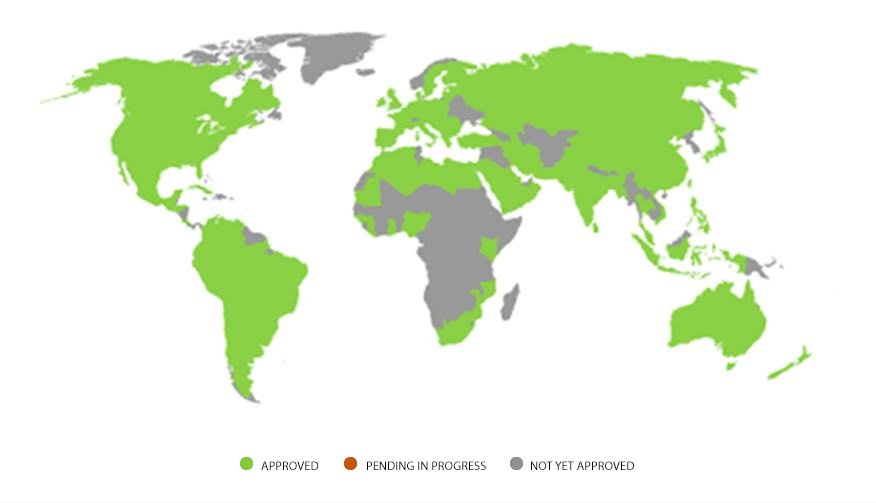
| AMERICAS | APAC | EUROPE/ME | AFRICA |
|---|---|---|---|
| United States; | Australia | Belarus | Algeria |
| Argentina | Bangladesh | Bahrain | Cape Verde |
| Bolivia | China | European Union | Egypt |
| Brazil | Hong Kong | Iran | Equatorial Guinea |
| Canada | India | Jordan | Gambia |
| Chile | Indonesia | Kazakhstan | Ghana |
| Columbia | Japan | Kuwait | Guinea Bissau |
| Costa Rica | Korea | Lebanon | Guinea Conakry |
| Ecuador | Malaysia | Omar | Kenya |
| Elsalvador | Myanmar | Qatar | Liberia |
| Guatemala | New Zealand | Russia | Libya |
| Mexico | Pakistan | Saudi Arabia | Morocco |
| Paraguay | Philippines | Switzerland | Maurutania |
| Peru | Singapore | Turkey | Nigeria |
| Uruguay | Sri Lanka | Uzbekistan | Sierra leone |
| Venezuela | Taiwan | Yemen | South Africa |
| Honduras | Thailand | Tunisia | |
| Vietnam | |||
| Source: Global Stevia Institute | |||
